With each new Apple Watch iteration, Apple has integrated additional health sensors. Over time, the device has gained the ability to track temperature changes and blood oxygen saturation, a feature that recently landed the company in hot water. In recent years, it has also had an electrocardiogram (ECG or EKG) sensor, which has been around since the introduction of the Apple Watch Series 4.
The EKG abbreviation is used most commonly in the United States. Apple dubbed their feature its ECG sensor, the common abbreviation in other English-speaking countries, so we'll refer to ECG moving forward. This sensor allows you to take an ECG reading right from your wrist, and share the results with your doctor or other health professional. Here we explain everything you need to know about the Apple Watch's heart features, how they work, what they mean, and why you might want to use them.
What are the Apple Watch heart health features?
- ECG/EKG
- Heart rate notifications
- Irregular rhythm notification
Apple first announced its ECG and irregular rhythm notification features in Sept. 2018, with the ECG feature specific to the Apple Watch Series 4 at the time. As you might expect, it's also available on later models: Watch Series 5, Watch Series 6, Watch Series 7, Watch Series 8, Watch Series 9, and the Watch Ultra and Watch Ultra 2. You won't find the ECG feature on the first or second-generation of the Watch SE though.
The ECG feature allows users to perform the tests from the comfort of their homes rather than requiring a local doctor's office or hospital to take a reading. For those not familiar with the term, an ECG is a way of measuring the timing and strength of electrical signals that make up your heartbeat. By reading an ECG, a doctor can see your heart's rhythm and any irregularities, intervening if required.
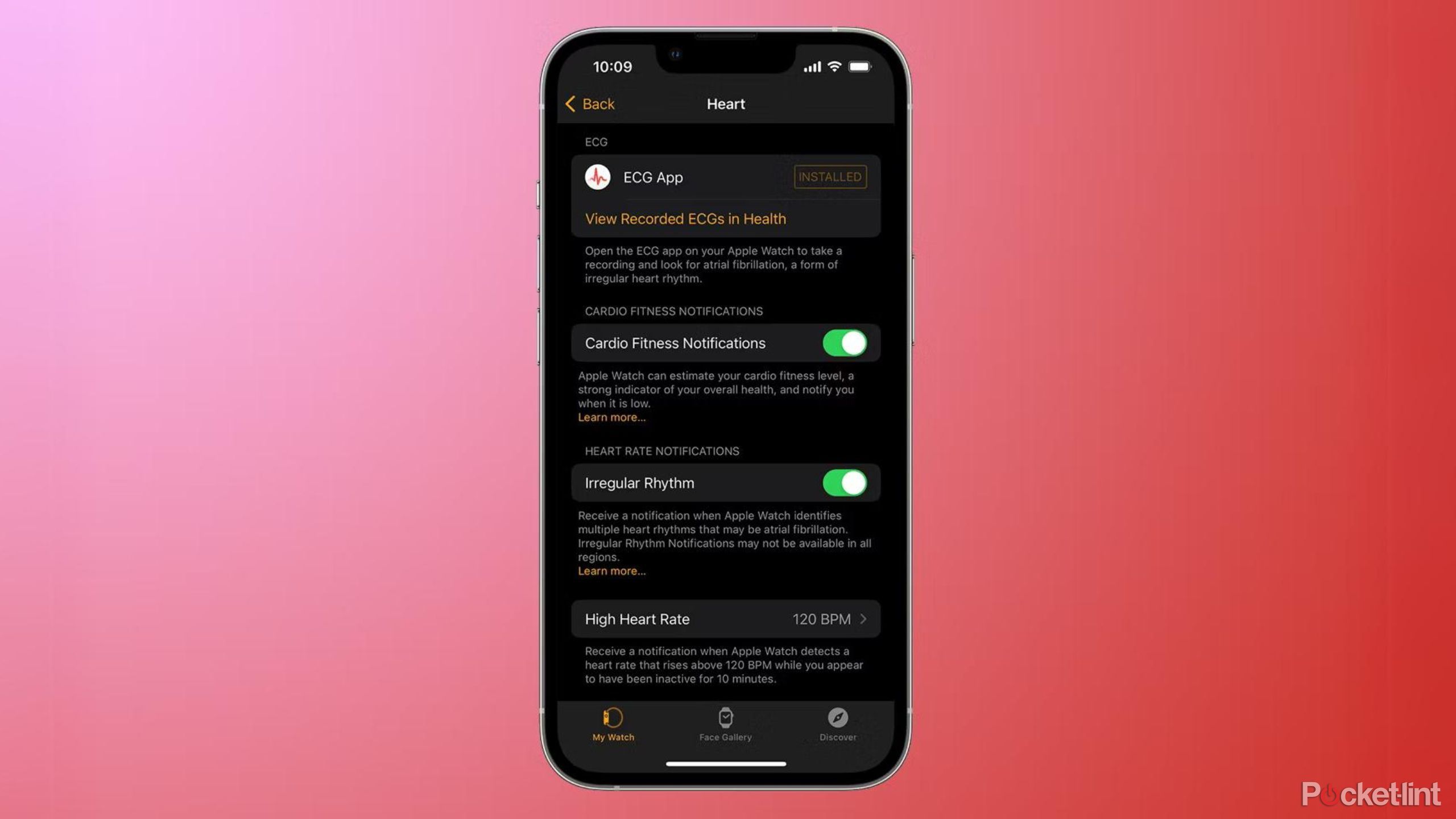
Meanwhile, the irregular rhythm notification feature sees the Apple Watch monitor your heart rhythm in the background every two hours and send a notification if it detects an irregular rhythm. It is available to all Apple Watch users with a Series 1 or later, including the Watch SE and Watch SE (second-generation).
Heart rate notifications can alert you if you have an unusually high or unusually low heart rate. If your heart rate is over 120bpm when your watch detects that you've been inactive for 10 minutes, you'll get a notification informing you of the fact. You'll also receive a notification if your heart rate is below 40bpm. You can adjust these thresholds if needed, or turn the alerts of completely if you wish.
How to get ECG on Apple Watch and which models have it
- Apple Watch Series 4, 5, 6, 7, 8, 9, Ultra, or Ultra 2 required for ECG
The ECG and irregular rhythm notification features are available for Apple Watches running Watch OS 5.2 or later. The irregular rhythm notification feature is available for all Apple Watch models from Series 1 and newer, including the Apple Watch SE models.
The ECG feature, meanwhile, requires the electrical heart rate sensor within the Digital Crown, which is unique to the Series 4, Series 5, Series 6, Series 7, Series 8, Series 8, Watch Ultra, and Watch Ultra 2 models.
What do you need to take an ECG with Apple Watch?
- ECG app
- Apple Watch Series 4, 5, 6, 7, 8, 9, Watch Ultra, or Watch Ultra 2
- Supported country
The ECG app is available in over 150 countries globally. Aside from the app, you'll also need the Apple Watch Series 4, Series 5, Series 6, Series 7, Series 8, Series 8, Watch Ultra, or Watch Ultra 2, an iPhone (iPhone 5 or above) and the ability to sit still while you do the test.
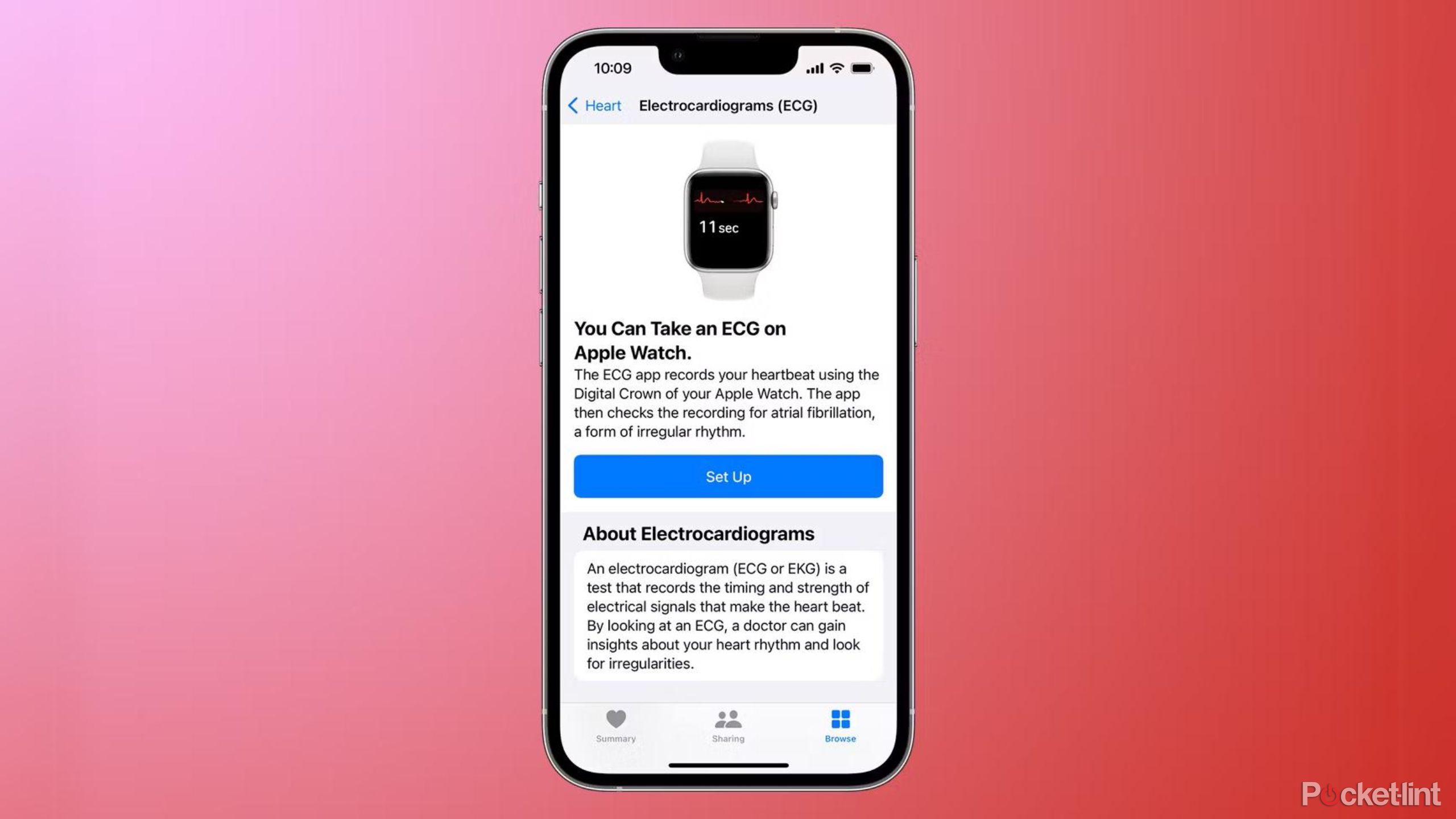
How to set up the Apple Watch ECG app
- iOS 12.2 and WatchOS 5.2 or later
- Requires iPhone Health app
In order to set up the Apple Watch ECG app, you'll need to make sure you're running iOS 12.2 or later on the iPhone connected to your Apple Watch and WatchOS 5.2 or later on your Apple Watch, after which you can access the ECG functionality in the Health app on your iPhone.
If you're doing this for the first time, you should see a prompt asking you to set it up. If you don't, you'll need to take the following steps.
- Open the Health app on your iPhone.
- Tap the Browse tab at the bottom right of your screen.
- Select Heart.
- Tap Electrocardiogram (ECG).
- Select Setup the ECG app.
Setup is incredibly easy and only requires a few details, such as your age. That's because performing an ECG under the age of 22 isn't recommended. Once set up, you can open the Apple Watch ECG app and begin taking an ECG reading.
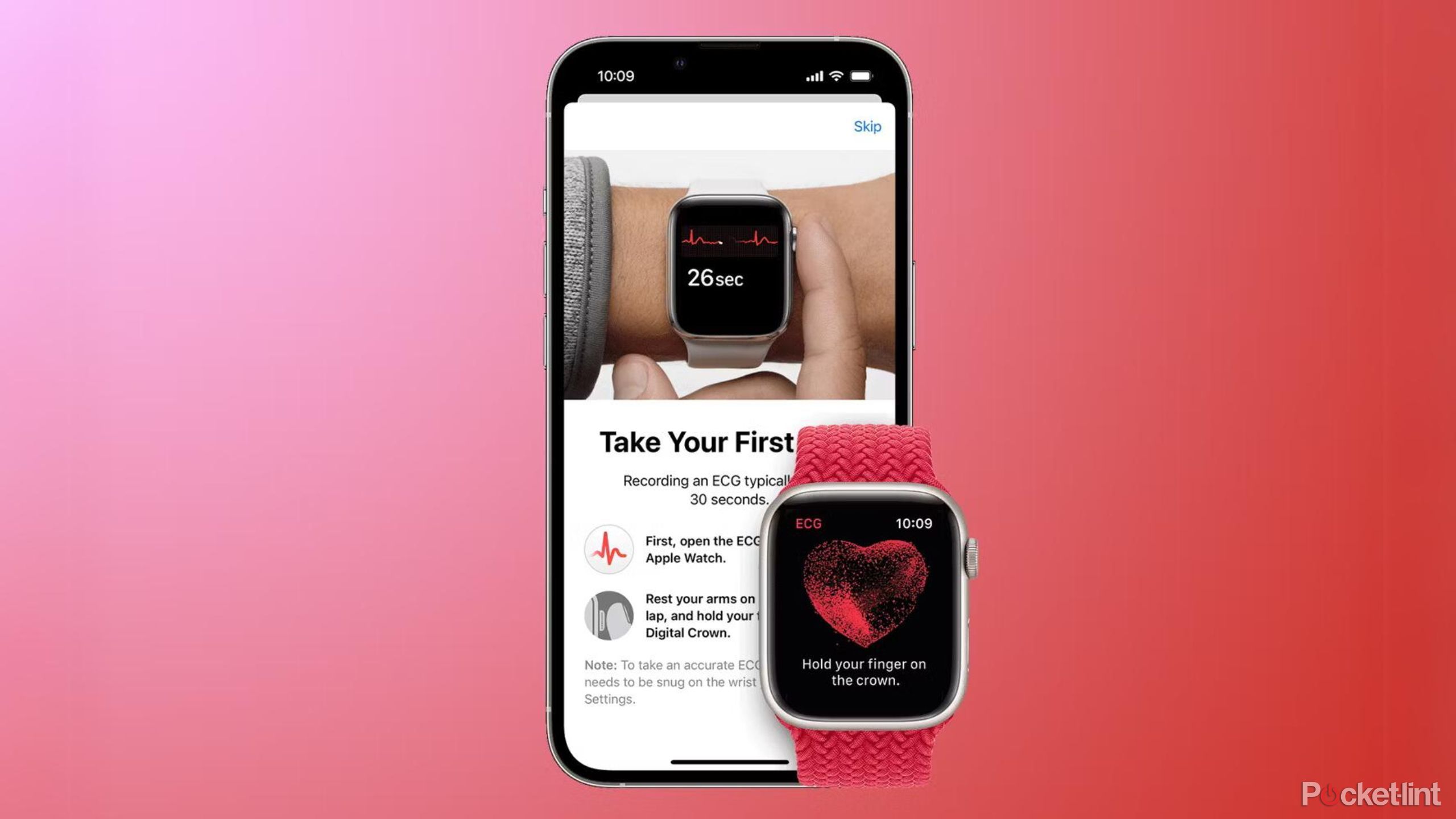
How to take an ECG on the Apple Watch
- Open ECG app on Apple Watch Series 4, 5, 6, 7, 8, 9, Ultra, or Ultra 2
- Hold your index finger on the Digital Crown
- Wait 30 seconds
Once you've set up the app, you can take an ECG whenever and wherever you want. The beauty of having the device on you all the time is that you don't have to book an appointment with the doctor.
Simply open the ECG app on the Watch (the icon with the image of a heart rate graph), rest your arm on something to reduce movement, and then hold your index finger on the Digital Crown without depressing it. It's all about making a looped connection for the system to monitor what's happening.
The test takes around 30 seconds to complete, and you'll see a representation of your heart rate on the screen while you do it. When complete, the Apple Watch will give you an instant result, along with the ability to record any symptoms you were feeling at the time. The text on your Watch makes clear that the ECG app can't detect signs of a heart attack.
On your iPhone, you'll get a notification with a link straight to the report, so you can see further details or share the results with your doctor.
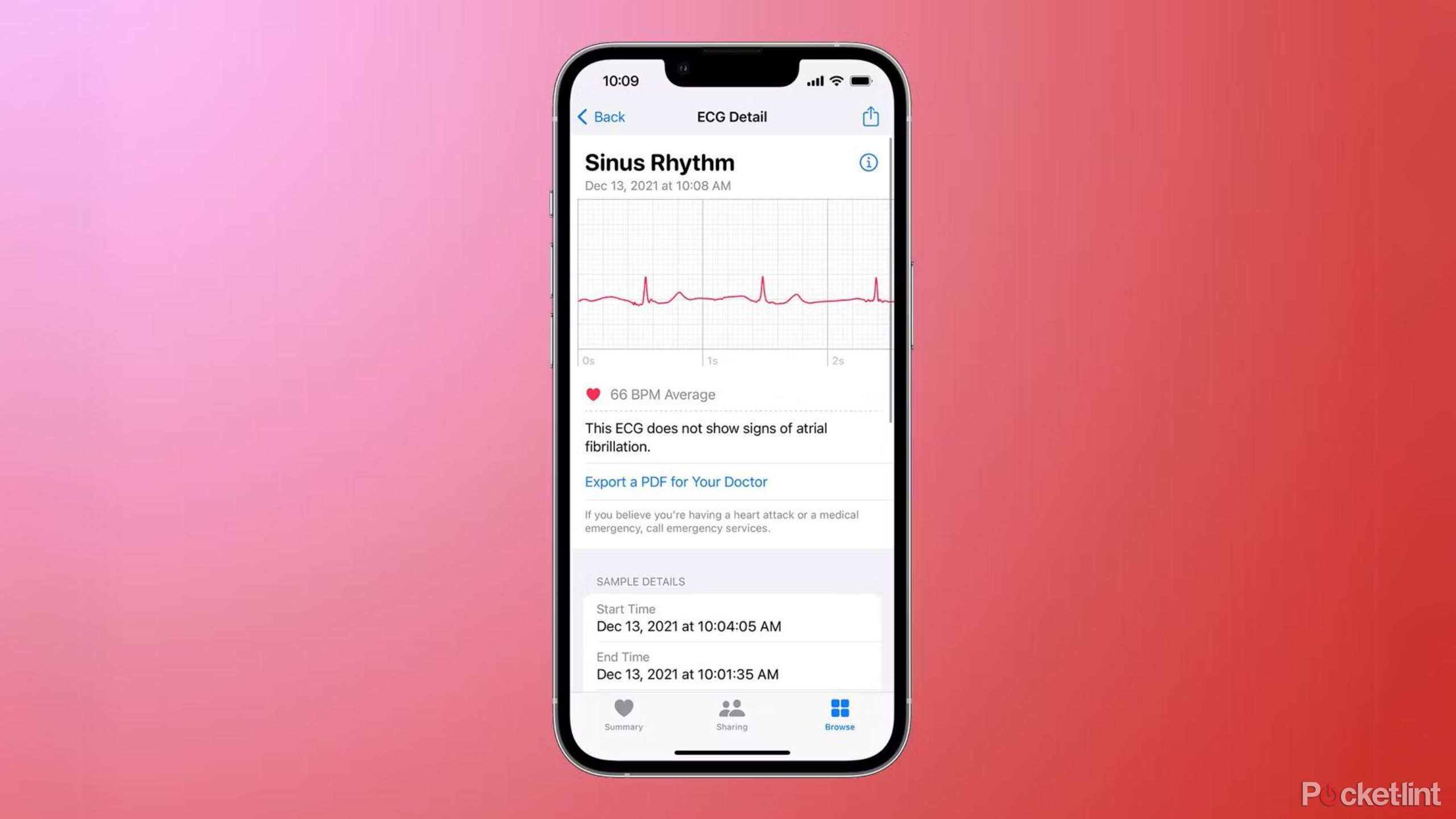
What do the Apple Watch ECG results mean?
Once you've finished capturing your EGC reading, you'll get one of four messages.
Sinus rhythm means everything is as expected. Atrial fibrillation means there is an irregular pattern detected. Low or high heart rate is the third potential result, while Inconclusive means the test can't determine the end result.
Can you share your health information with your doctor?
It's easy to share your ECG results with a health professional if you have any abnormal results.
- Open the Health app on your iPhone.
- Select Browse.
- Tap Heart.
- Select Electrocardiogram (ECG).
- Tap ECG result.
You can then export the details via a PDF to share with your doctor. You decide how that information is shared. Apple doesn't share that information with anyone else, such as third-party insurance companies, other apps, or even your own computer.
 Is it as good as an ECG in a doctor's office?
Is it as good as an ECG in a doctor's office?
Nobody is saying the Apple Watch ECG is as good as or capable of replacing a medical-grade electrocardiograph machine, but that's not the idea. A traditional hospital ECG is often referred to as a 12-lead machine because it uses 10 different electrodes to provide information on 12 different areas of the heart. In contrast, the Apple Watch is akin to a single lead machine.
The documentation refers to the Apple Watch ECG not being a substitute for actual medical care at every opportunity. Support documents from Apple state that the "user is not intended to interpret or take clinical action based on the device output without consultation of a qualified healthcare professional." Furthermore, "the feature is not intended to replace traditional methods of diagnosis or treatment".
To get the device cleared by the relevant regulatory bodies in the US and Europe, Apple had to provide details of a clinical test of around 600 people, half of whom had atrial fibrillation. The ECG app was unable to read about 10% of the subjects, but for the others, it was very accurate, according to Apple. The company said "it caught more than 98% of people with atrial fibrillation, and correctly told people that they didn't have the condition 99.6 percent of the time."
Apple also carried out another substantial test involving the accuracy of Apple Watch's heart data -- the Apple Heart Study with Stanford University, for example, monitored atrial fibrillation, but not the ECG functionality.
Should Apple give people the ability to do ECG readings at home?
Apple has clearly been encouraged by Apple Watch's various successes as a personal medical device. Indeed, it's done a better job at this than any other device before it. The detection of high heart rates has been especially popular and there have been a few cases where the Apple Watch has probably saved lives because it has prompted people to seek medical help.
Ivor Benjamin, president of the American Heart Association, was on stage with Apple when the company announced the feature in September 2018. "In my experience, people often report symptoms that are absent during their medical visits," Benjamin said. Then adding, "[This] is game-changing, especially when evaluating atrial fibrillation -- an irregular and rapid heart rate that can increase a person's risk of stroke, heart failure and other heart-related complications.
In the UK, Professor Martin Cowie, professor of Cardiology at Imperial College London, based at Royal Brompton Hospital, and chair of the Digital Health Committee of the European Society of Cardiology, also was encouraged by the ECG feature's capabilities when it arrived.
Today, there are around 1.5 million people in the UK living with atrial fibrillation, the most common heart rhythm problem, but a third of these people may be unaware of this. An on-demand ECG and pulse check could be a powerful tool in our ongoing quest to manage heart health better across Europe. The opportunity for innovation to optimize patient care is huge and this is a great step forward.
What is atrial fibrillation?
Atrial fibrillation (or Afib/AF) is where the upper chambers of the heart beat irregularly, increasing the risk of stroke and heart failure. The irregular rhythm notification feature will check heart rhythms every two hours in the background and send a notification if an irregular heart rhythm is detected.
If an irregular heart rhythm is detected, the Apple Watch will do five further tests in quick succession to see if the outcome is still positive. If it is positive, those using an Apple Watch Series 4, 5, 6, 7, 8, 9, Watch Ultra, or Watch Ultra 2 device will be prompted to do an ECG reading. Those using an Apple Watch Series 1, 2, 3 or either Watch SE model will suggest you contact your local GP.
 Which countries does Apple Watch ECG work in?
Which countries does Apple Watch ECG work in?
Rather than roll out the ECG feature to all Apple Watch users, Apple wanted it to only be available in regions "cleared" for a medical device. Because of that, the ECG feature wasn't initially available worldwide, though it's pretty much there now.
Apple's ECG feature is available in the US, UK, and over 150 other regions and countries globally. You can see the full list of supported countries here.
What can't the Apple Watch heart health features do?
The Apple Watch heart features do not detect a heart attack, blood clots, a stroke or other heart-related conditions including high blood pressure, congestive heart failure, high cholesterol or other forms of arrhythmia.
What are the risks?
There are a few caveats to the heart features on Apple Watch mentioned in the FDA ruling on the device, not least that "the ECG app is not intended for use by people under 22 years old [or…] individuals previously diagnosed with AFib." You also need to be still for the ECG to work. Equally, "it is not intended to provide a notification on every episode of irregular rhythm suggestive of AFib and the absence of a notification is not intended to indicate no disease process is present." It's a guide, not a 99.9% accurate test.
That's key, because some will feel completely assured by the ECG app and not seek appropriate care: "Over-reliance on device output leading to failure to seek treatment despite acute symptoms or discontinuing or modifying treatment for chronic heart condition". The FDA ruling also points out that "false positive resulting in additional unnecessary medical procedures" could be an issue with the device, resulting in people going for actual ECGs that don't need to happen.